Covid vaccine winners set sights on flu as their next big target
After having beaten established vaccine makers to produce the first Covid-19 shots, Moderna and BioNTech, with its partner Pfizer, are racing to dethrone them in another big market: flu.
They are betting that the new messenger RNA technology used in their Covid vaccines at large-scale for the first time, will eventually allow flu jabs to be developed more quickly to match ever-changing annual strains and improve efficacy rates of between 40 and 70 per cent.
The challengers have also forced incumbents such as Sanofi, Seqirus and GlaxoSmithKline, to speed up their research into mRNA alternatives, even though they argue their existing products may still prove superior.
They have a lot to lose if they fall behind. According to research group Fortune Business Insights, this already large market is forecast to grow at 7.2 per cent a year to $10.7bn in 2028. Sanofi predicts it could be even bigger, hitting $17bn by 2030.
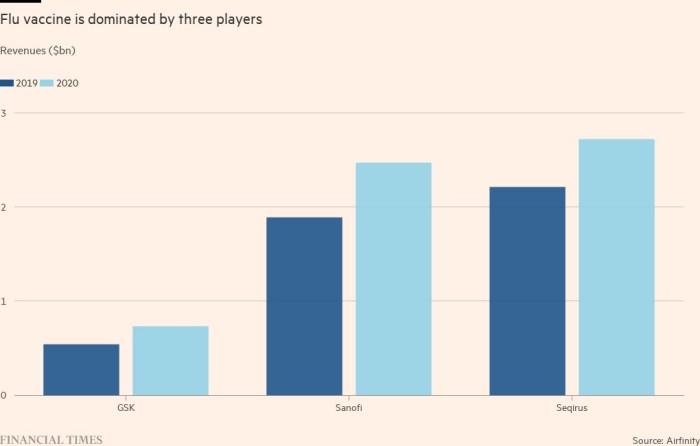
As people became more aware of the threat of respiratory viruses — and the potential of vaccines to protect them — revenue at the top three flu vaccine makers grew by 23 to 35 per cent from 2019 to 2020. About 40 per cent of Sanofi’s €6bn in vaccine revenues last year came from flu jabs.
Moderna shook up the race last week when it announced early data showing its flu vaccine candidate boosted antibody levels in a phase 1 trial, even for participants receiving the lowest dose. Pfizer and BioNTech started enrolling participants in their phase 1 study in September.
Stephen Hoge, Moderna’s president, told investors that the vaccine candidate, which elicited roughly the same immune response as Sanofi’s US market leader Fluzone, was just a “starting point”.
He is a “big believer” in the potential advantages of combining vaccines for flu and Covid in a single shot. “We want to be as good as we’re seeing with enhanced vaccines,” he said. “And then what we want to do is combinations.”
“If [mRNA] does work, you could cede the whole market to Moderna or Pfizer,” said Geoffrey Porges, an analyst at healthcare-focused investment bank SVB Leerink.
Every year, the World Health Organization has to predict which flu strains will be circulating in the next winter. With current flu vaccines, they make this choice as early as February.
But mRNA manufacturing, which entails inserting a genetic code for part of the virus in a bubble of fat, is far faster than older technologies that grow active ingredients in tanks.
Hoge told the Financial Times that this could allow a new vaccine for the winter to be designed as late as the summer, making it “not just an educated guess”, because there would be more data on the strains circulating in the southern hemisphere winter.
He said this potential advantage means the opportunity for Moderna in the flu market is just as significant as for regular Covid-19 boosters after the pandemic.
“The current flu efficacy rates are terrible,” he added.
Eventually, mRNA could be used to combine a vaccine against the four annual strains of flu prioritised by the WHO, the current variant of Sars CoV-2 and respiratory syncytial virus, which can be serious for infants and the elderly. Moderna has already shown it can pack six different mRNA sequences in one dose, in a vaccine being trialled for cytomegalovirus, which can cause problems in babies and young children.
“We think people don’t want three boosters. One injection for pan-respiratory protection feels like the right answer,” Hoge said.
The flexibility of mRNA might also mean it could eventually allow vaccines to be updated for extra strains during flu season, or could be used to create different regional vaccines.
Mikael Dolsten, Pfizer’s chief scientific officer, told investors that the US drugmaker is “very intrigued by the use of mRNA for flu” because it can generate a good response from T cells, white blood cells that are a part of longer-term immune protection, improving on current vaccines.
CureVac, a German biotech that has yet to have a Covid vaccine approved, is also hoping to enter the flu market, with its partner UK drugmaker GlaxoSmithKline.
Franz-Werner Haas, CureVac’s chief executive, said artificial intelligence could help predict future evolutions of the strain.
GSK had not been investing heavily in its flu business before the pandemic because it was dependent on the traditional method of growing the active ingredient in eggs. Now, Roger Connor, GSK’s president of vaccines, said flu was in the “sweet spot” for mRNA, where current vaccines have such low efficacy that it is “effectively an unmet need”.
“MRNA is something we’re investing in significantly at the minute. We absolutely intend to be a leader,” he said.
CureVac and GSK have yet to start clinical trials but Haas said just because the Covid vaccine makers are out in front now, it does not mean they will have the first approved products for flu.
“It’s a race and hopefully for the benefit of all,” he said.
But while Moderna’s phase 1 study showed its vaccine was safe and produced a robust antibody response, making it confident to proceed to the next stage of trials, some investors were disappointed.
In a separate study, its vaccine candidate induced similar levels of antibodies to Sanofi’s Fluzone, the highest dose shot, which is targeted at older adults. Moderna shares dropped 5.6 per cent on the day it announced the data, while Sanofi rose 3 per cent.
Thomas Triomphe, who leads research and development at Sanofi’s vaccine business, said the results showed what the company has always said: that “flu is not Covid” — that flu vaccines need to be easy to distribute and have very few side effects.
“What’s important is the current generation of mRNA . . . is a great pandemic generation but does not have the proper fit outside of the pandemic market,” he said.

Jo Walton, an analyst at Credit Suisse, said there was a “much higher hurdle for safety in mRNA for flu” than for Covid, partly because deaths from flu often result from cardiac complications and the mRNA Covid vaccines have been associated with rare cardiac side effects.
“The last thing you want to do is have any implication of cardiac issues in side effects, so the safety study scrutiny will be pretty intense,” she said.
Sanofi’s flagship flu vaccine has been shown to protect against serious complications from flu, including heart attacks. An mRNA vaccine may not be able to prove this added benefit — or it may take time to collect that data.
Ethan Settembre, vice-president of research and development at Seqirus, said it was only “early days” in our understanding of how well mRNA will work for flu and improve on the “quite good” existing vaccines.
“We are always searching to make better flu vaccines,” he said. “And new technologies sometimes provide that option. Sometimes they’re right, sometimes they’re not right.”
While work has already been done to make the Covid mRNA vaccines easier to store and transport, they also require more careful handling than flu vaccines, which are often given in pharmacies or doctors’ offices because they do not need the same level of cold storage.
Seqirus is working on an improved version of its protein-based vaccine, which is grown in cells, not eggs, and uses an adjuvant to boost immune response. He said that will probably have an advantage in stability and is proved to elicit robust responses from those most vulnerable to flu, children and the elderly.
But it is also developing an mRNA vaccine, using a novel self-replicating technology that amplifies its effect, which is scheduled to begin human trials in the second half of next year.
Sanofi meanwhile has already launched a phase 1 trial of an mRNA flu vaccine. Triomphe believes the company is well placed to succeed: it bought mRNA start-up Translate Bio for $3bn earlier this year, and has promised to plough €400m a year into mRNA vaccine research with 400 scientists working in the field.
The French company hopes that this, combined with its relationship with institutional buyers and government bodies that issue guidance on flu vaccines, will help it beat back the threat from Pfizer, BioNTech and Moderna.
Credit Suisse’s Walton believes that though the market is undoubtedly changing, it will take a lot to shift the advantages of incumbency. “If Sanofi has at worst a three-year delay, best case, a one-year delay after the mRNA vaccine makers, is that enough to kill their franchise or not? I suspect it isn’t.”